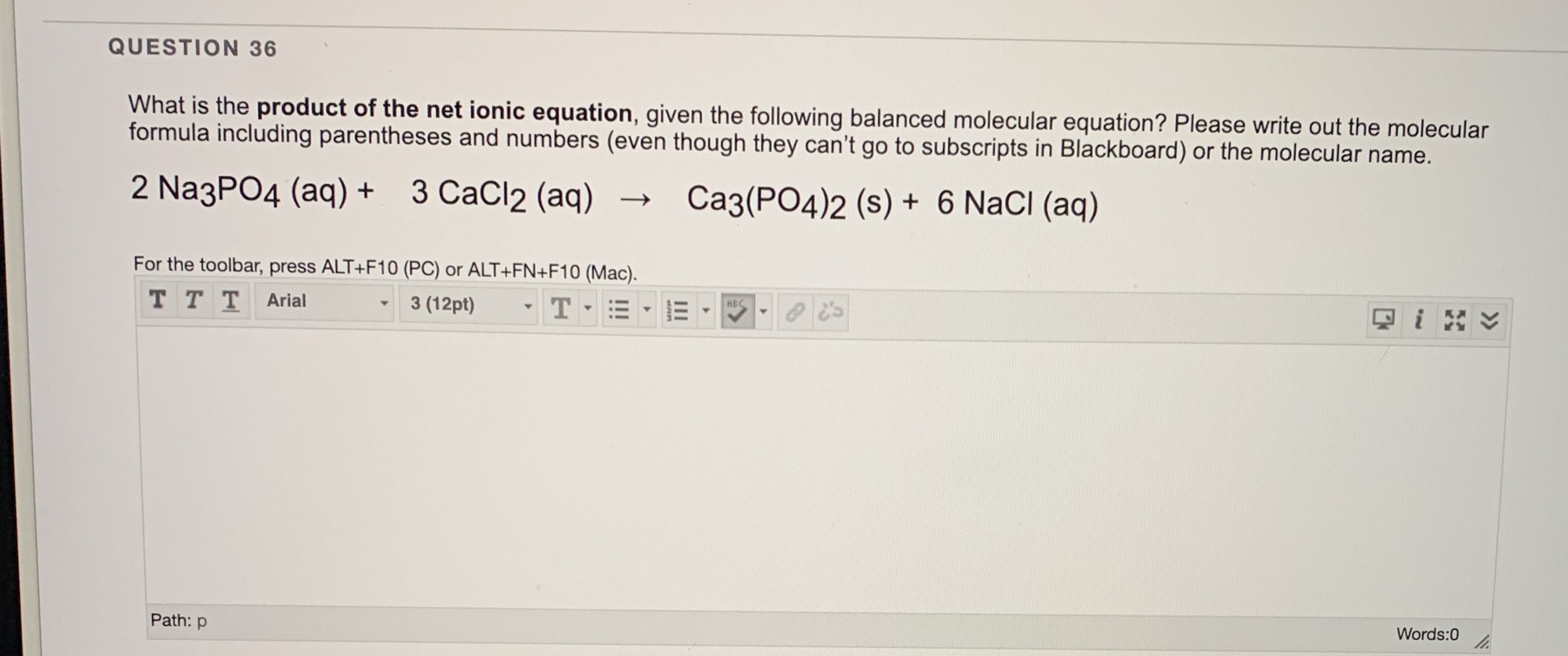
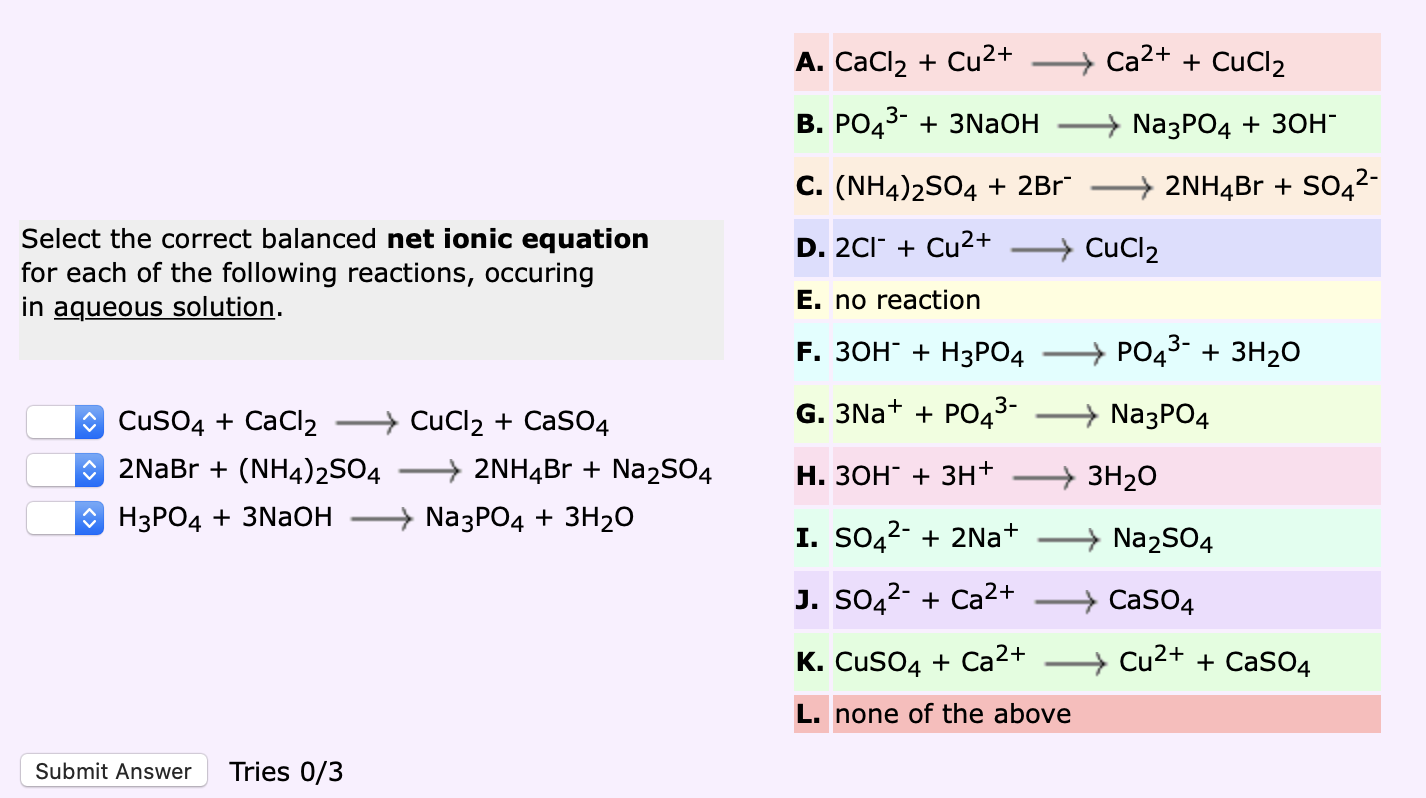
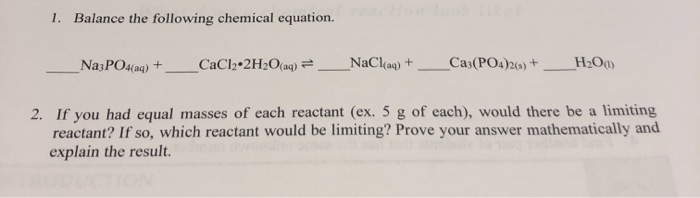SOLVED:Please express the following chemical reactions as their chemical equilibrium formula. Give the balanced equations and give the chemical equilibrium formula_ 1.) AgNO3(aq) K2CrO4(aq) = Ag2NO3 KNO3 2.) CuCl2(aq) Pb(NO3)2(aq) Cu(NO3)2 PbCi2

Chemical Equations Chapter 11 Notes. Balanced Equation 2 NaCl + F 2 2 NaF + Cl 2 ReactantsProducts Yields is the “equals” Coefficient used to balance. - ppt download